ISMPP Academy report: Oxford PharmaGenesis teaches how to elevate publications through enhanced impact and demystifies RWE and HEOR
19 October 2023
Share this page
More than 150 medical publications professionals gathered in Philadelphia on 28–29 September 2023 for the inaugural ISMPP Academy. Oxford PharmaGenesis was a key event sponsor and we were delighted to give two valuable presentations to help attendees to explore new frontiers in publications.
In line with the ISMPP Academy theme ‘PREP! Practical Resources to Elevate Publications’, Oxford PharmaGenesis colleagues Richard White (Chief Operating Officer) and Tomas Rees (Director of Innovation), along with Leah Bensimon (Director, Scientific Publications at Bristol Myers Squibb) presented talks covering new ways of thinking about publication impact, and explaining the basics of real-world evidence (RWE) and health economics and outcomes research (HEOR).
If you missed the talks or need a reminder of the content, here are some of the key takeaways from both.
New ways of thinking about impact
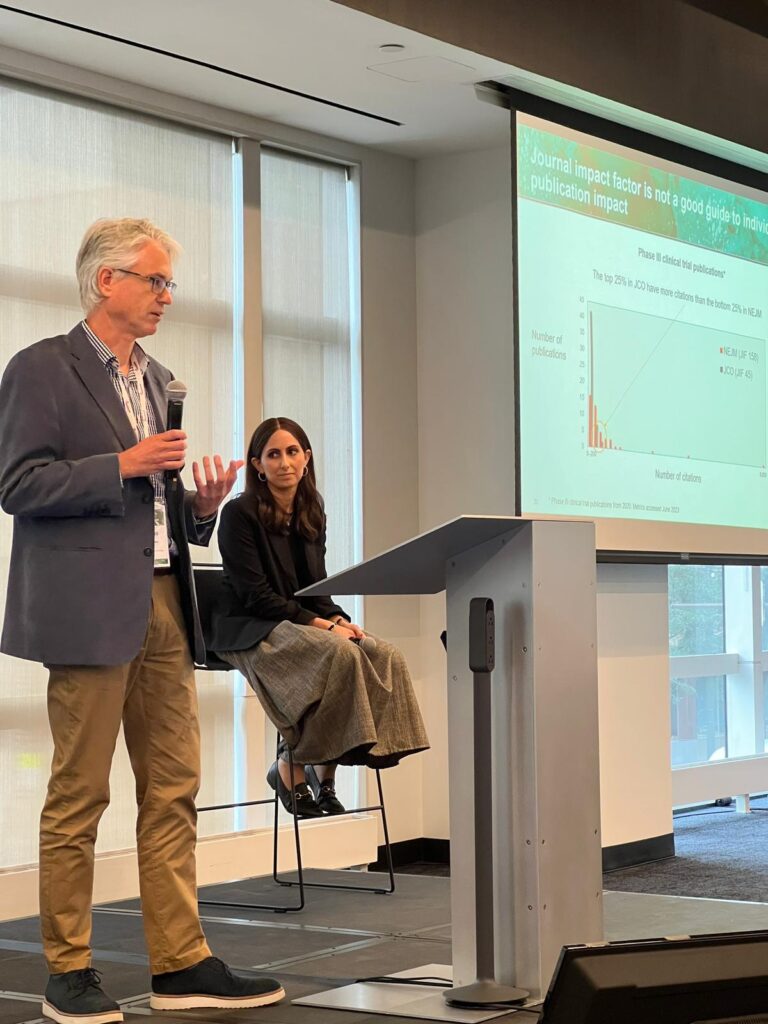
In their talk ‘New ways of thinking about impact’, Tomas Rees and Leah Bensimon explored the best way to break away from traditional, linear publications planning and embrace different options to expand reach.
The talk promised to help attendees to apply new strategies to increase the reach and impact of publications plans, identify the best methods for analytics and collecting metrics, demonstrate value when requesting more resources, and more.
Presenters Tomas and Leah described why we need metrics, explaining that they help us to get a better understanding of the value of publications and how they contribute to ethical commitments, support product launches, shape treatment practice and advance the science.
They then looked at the kind of insights needed to demonstrate value, exploring types of publications and how they are used by the field team for medical information and market access and so on.
Tomas and Leah introduced ways to measure impact and outlined the difference between quantitative and qualitative metrics. They then addressed how to use article-level metrics to evaluate different kinds of impact and went on to explore how using metrics across articles at scale can provide a variety of insights, such as level-setting and prediction, gap analysis and areas of research activity.
Tomas looked at using AI techniques to evaluate the literature, explore topics and obtain qualitative insights. He said AI can help us analyse large amounts of data and find patterns, trends and relationships that we might otherwise miss. He gave some examples of how AI can help us with topic modelling, sentiment analysis, text summarization and more.
They also presented ways to think about increasing the impact of publications, such as publication enrichments and extensions, and explored some of the opportunities and barriers to using social media to communicate publications.
The session finished with case studies. Tomas presented work carried out with Ipsen that benchmarked and assessed all their publications so that the company could see impact levels across products, publication types and other publication characteristics. Leah presented a case study using sentiment analysis social media discussions to assess the impact of a publication addressing potential safety concerns.
The basics of RWE and HEOR for publications professionals

With the fields of RWE and HEOR changing on an almost daily basis, Richard White’s presentation at the ISMPP Academy provided a timely overview of the field and an update for publications professionals on the latest guidance from the Food and Drug Administration (FDA), Professional Society for Health Economics and Outcomes Research (ISPOR)-International Society for Pharmacoepidemiology (ISPE) and other groups on best practices for the planning, design, execution and communication of RWE and HEOR studies. A packed session with 52 attendees and standing-room only attested to the continued interest of ISMPP members in the application of RWE and HEOR in clinical decision-making.
Richard opened by emphasizing the importance of understanding and respecting the ‘why’ of RWE and HEOR teams – their perspectives, work and goals – in order to build a collaborative working relationship. Best practice should include a joint RWE/HEOR/medical/publications stakeholder workshop to co-create communications plans and build contingencies, ensuring compliance with Good Publication Practice (GPP) and agreement on preferred document templates and writing guidelines/checklists.
In the second part of the session, Richard focused on the inherent differences between RWE studies and RCTs, including issues such as complex stakeholder involvement, uncertainty over data sufficiency, fundamental limitations of the data and target journal selection. This was followed by a summary of external challenges to the credibility of industry-sponsored RWE and HEOR studies, and how trust in these can be built through transparency by using newly established reporting registries, guidelines and checklists.
Finally, Richard encouraged attendees to add value through their own medical communications expertise. Publication enhancements, plain language summaries (PLS), animations and videos, and plain language summaries of publications (PLSPs) are hugely important tools that should be used to explain RWE and HEOR studies, because this is an area in which most healthcare professional and patient audiences have little basic understanding.
The session included an opportunity for audience questions, which went beyond the session and into the lunch-break. Topics discussed included the following.
- The challenge of modelling health benefits over a lifetime when health insurance companies consider a much shorter budget horizon.
- The complexities of determining authorship of HEOR studies and when clinical experts should be involved to meet ICMJE criteria.
- Requests from journals for access to RWE study data when such data are owned by a third-party vendor.
- The growing number of methodologies to correct for bias in RWE studies, such as propensity score matching, matching-adjusted indirect comparison (MAIC), inverse treatment probability weighting (ITPW) and others.

