 Oxford PharmaGenesis this week expanded its real-world evidence experience into the areas of respiratory and infectious diseases, with the commissioning of two new SURE™ (Systematic Understanding of Real-World Evidence) assessments. This brings to double digits the number of SURE™ projects initiated in the past 12 months, which have covered cardiology, neurology, diabetes, endocrinology, oncology, immunology and nephrology. We have also conducted SURE™ assessments in several orphan indications.
Oxford PharmaGenesis this week expanded its real-world evidence experience into the areas of respiratory and infectious diseases, with the commissioning of two new SURE™ (Systematic Understanding of Real-World Evidence) assessments. This brings to double digits the number of SURE™ projects initiated in the past 12 months, which have covered cardiology, neurology, diabetes, endocrinology, oncology, immunology and nephrology. We have also conducted SURE™ assessments in several orphan indications.
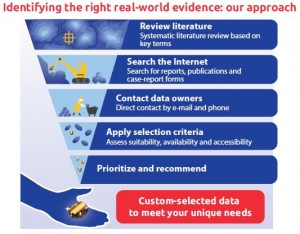
SURE™ assessments are a unique service developed by Oxford PharmaGenesis that employs a targeted, systematic and comprehensive methodology to identify, compile, assess and recommend data sources for observational research. SURE™ projects routinely assess diverse real-world data sources such as registries, administrative healthcare databases, hospital discharge data, electronic health records and epidemiology cohorts, and have also included specialized data elements (e.g. inpatient drugs, laboratory and imaging results, functional status, patient-reported outcomes, tumour stage) where data linkage maybe required.
“It is thrilling to see demand for our SURE™ assessments across such a wide range of therapy areas,” said Dr Richard White, Commercial Director. “Real-world evidence is increasingly informing decisions that influence product approval, reimbursement and uptake, and our clients want our help in identifying relevant, high-quality and accessible data sources for their needs. A clear illustration of the value of our unique methodology is that several of our clients have designed their observational study programmes based on our SURE™ assessments.”
Find out more about our SURE™ methodology and our real-world evidence capabilities by visiting www.pharmagenesis.com/healthcare-medical-services/real-world-evidence or by visiting our exhibition booths at TIPPA (29 February–1 March), ISMPP (11–13 April) or ISPOR (21–25 May).
